我的博文
2020年WHO分类软组织肿瘤病理学新进展(1)
The 2020 WHO Classification What’s New in Soft Tissue Tumor Pathology?
The new fifth edition of the World Health Organization (WHO) Classification of Tumors of Soft Tissue and Bone was published in early 2020, 7 years following publication of the fourth edition. The revisions reflect a consensus among an international expert editorial board composed of soft tissue and bone pathologists, geneticists, a medical oncologist, surgeon, and radiologist. Updates to the classification reflect extensive new genetic data, prompting expansion and reorganization of several categories. The soft tissue tumor chapter in particular includes the addition of recently described tumor types; in some cases, increasing outcome data and comparative genetic studies argued for splitting into separate tumor types or changes in nomenclature and managerial categories. Despite the expanding contribution of genetics to our understanding of the molecular pathogenetic basis for soft tissue tumors, the new classification emphasizes the continued central diagnostic importance of morphology. | 世界卫生组织(WHO)新的第五版软组织和骨肿瘤分类在第四版出版7年后于2020年初出版。修订反映了一个国际专家编辑委员会的共识,该委员会由软组织和骨病理学家、遗传学家、医学肿瘤学家、外科医生和放射科医生组成。对分类的更新反映了大量新的遗传数据,促进了几个类别的扩展和重组。软组织肿瘤一章特别包括最近描述的肿瘤类型的增加;在某些情况下,越来越多的结果数据和比较遗传学研究主张分裂成不同的肿瘤类型或改变命名和管理类别。尽管遗传学对我们理解软组织肿瘤的分子病因基础的贡献不断扩大,但新的分类强调持续关键的形态学诊断的重要性。 |
In addition to a discussion of “new” soft tissue tumor entities, this review will highlight updated prognostic information for familiar tumor types, such as dedifferentiated liposarcoma (DDLPS) and solitary fibrous tumor (SFT). Finally, we will include a focused discussion of the dizzying array of recently described genetic alterations in soft tissue tumors. The past decade has seen a plethora of novel gene fusions in rare soft tissue tumor types; a comprehensive and in depth examination of these alterations is beyond the scope of this review. Instead, we will focus on selected genetic alterations with practical diagnostic relevance. An example is the undifferentiated small round cell sarcoma chapter; detection of novel gene fusions has led to subclassification of the former wastebasket of “Ewing-like” sarcomas into new categories with distinct presentations, clinical behavior, and morphologic and immunohistochemical (IHC) features. Additional practical examples include the identification of recurrent genetic alterations in SFT and malignant peripheral nerve sheath tumor (MPNST), leading to the introduction of highly sensitive and specific IHC markers, which are now in routine use in surgical pathology. | 除了讨论“新”的软组织肿瘤实体外,这篇综述将着重介绍常见肿瘤类型的最新预后信息,例如去分化脂肪肉瘤(DDLP)和孤立性纤维性肿瘤(SFT)。最后,我们将集中讨论最近描述的一系列令人眩晕的软组织肿瘤的基因改变。在过去的十年里,在罕见的软组织肿瘤中发现了很多新的基因融合;对这些改变的全面而深入的研究超出了本综述的范围。相反,我们将把重点放在有实际诊断意义的特定基因改变上。如未分化小圆细胞肉瘤这一章;新基因融合的检测已经导致以前的“尤因样”肉瘤的废纸篓亚分类为新的类别,具有不同的表现、临床行为、形态学和免疫组化(IHC)特征。其他的实际例子包括识别SFT和恶性外周神经鞘瘤(MPNST)中的重现性基因改变,从而引入高度敏感和特异的IHC标记物,现在在外科病理学中常规使用这些标记物。 |
NEW SOFT TISSUE TUMOR TYPES | 新的软组织肿瘤类型 |
The fifth edition of the WHO Classification features “new” soft tissue tumor types that were not found in the fourth edition (Table 1). As was the case for prior editions, tumors are organized by line of differentiation, such as adipocytic, fibroblastic/myofibroblastic, vascular, and smooth muscle neoplasms. Several emerging tumor types, including the morphologically heterogenous NTRK-rearranged spindle cell neoplasms, are contained within the section on tumors of uncertain differentiation. Epithelioid hemangioendothelioma (EHE) with YAP1-TFE3 fusion will be discussed in the novel genetic changes section below. Also discussed here are “new” tumor types within the undifferentiated small round cell sarcoma chapter. | 世界卫生组织第五版分类的特点是具有第四版中没有发现的“新”软组织肿瘤类型(表1)。与以前的版本一样,肿瘤是按组织分化,如脂肪细胞、成纤维细胞/肌纤维母细胞、血管和平滑肌肿瘤。一些新出现的肿瘤类型,包括形态上异质的NTRK重排梭形细胞肿瘤,包含在不确定分化的肿瘤部分。伴YAP1-TFE3融合的上皮样血管内皮瘤(EHE)将在下面的新基因变化部分讨论。这里还讨论了“新”肿瘤类型内未分化小圆细胞肉瘤一章。 |
| |
Atypical Spindle Cell/Pleomorphic Lipomatous Tumor | 非典型梭形细胞/多形性脂肪瘤样肿瘤 |
Atypical spindle cell/pleomorphic lipomatous tumor is a benign adipocytic neoplasm, distinct from atypical lipomatous tumor/well-differentiated liposarcoma (ALT/WDLPS) and spindle cell/pleomorphic lipoma, although it shares some histologic features with both of these other tumor types. | 非典型梭形细胞/多形性脂肪瘤样肿瘤是一种良性脂肪细胞肿瘤,不同于非典型脂肪瘤样肿瘤/高分化脂肪肉瘤(ALT/WDLPS)和梭形细胞/多形性脂肪瘤,尽管它与其他两种肿瘤有一些共同的组织学特征。 |
The largest series of atypical spindle cell lipomatous tumors reported a male-to-female ratio of 3:2 and a median age of 54 years. | 最大病例数的一篇非典型梭形细胞脂肪瘤样肿瘤报道,男女比例为3:2,中位年龄为54岁。 |
Approximately two thirds of tumors occur in the limbs and limb girdles, with a predilection for the hands and feet, and an approximately equal distribution between superficial and deep sites. | 大约三分之二的肿瘤发生在四肢和肢带,好发于手和足,表浅和深部组织的分布大致相等。 |
Less commonly involved are the head and neck, genitals, and trunk, with very rare retroperitoneal involvement. | 较少发生于头颈部、生殖器和躯干,很少有腹膜后受累。 |
By contrast, ALT/WDLPS occurs equally commonly in the retroperitoneum and limbs, less commonly in the spermatic cord, mediastinum, and head and neck. | 相比之下,ALT/WDLPS均常见于腹膜后和四肢,较少出现在精索、纵隔和头颈部。 |
Spindle cell/pleomorphic lipomas characteristically occur in the posterior aspect of the neck or upper back, less often the face, orbital region, and oral cavity, with few reports of deepseated lesions in a broader anatomic distribution. | 梭形细胞/多形性脂肪瘤主要发生在颈后部或上背部,较少发生在面部、眼眶区和口腔,很少有更多的解剖分布的深部病变报道。 |
Histologically, atypical spindle cell lipomatous tumor shows infiltrative margins and demonstrates a range of appearances, with varying proportions of spindle cells, adipocytes, and lipoblasts, including hyperchromatic and sometimes pleomorphic cells (atypical pleomorphic lipomatous tumor). | 组织学上,非典型梭形细胞脂肪瘤样肿瘤表现为浸润性边缘,由一系列不同比例的梭形细胞、脂肪细胞和脂肪母细胞组成,包括深染细胞和有时多形性细胞(非典型多形性脂肪瘤样肿瘤)。 |
The histology may be viewed as a spectrum flanked by 2 morphologic extremes: a paucicellular pattern with bland spindle cells, minimal nuclear atypia, adipocytes, and abundant (often myxoid) matrix, and a hypercellular pattern at the other end of the spectrum with mild-to-moderate cytologic atypia, easily identifiable lipoblasts, and less abundant matrix (Fig. 1). | 组织学可以被看作是一个由两个极端形态组成的谱:细胞稀少模式表现为梭形细胞温和,核异型性最小,脂肪细胞和丰富的基质,通常是粘液样基质;另一个是富于细胞模式,细胞轻到中度异型性,易于识别的脂肪母细胞,以及不太丰富的基质(图1)。 |
The differential diagnosis includes spindle cell/pleomorphic lipoma and ALT/WDLPS. | 鉴别诊断包括梭形细胞/多形性脂肪瘤和ALT/WDLPS。 |
Spindle cell/pleomorphic lipomas have a different anatomic distribution, are well circumscribed, and contain characteristic brightly eosinophilic, coarse “ropy” collagen fibers. | 梭形细胞/多形性脂肪瘤具有不同的解剖分布,边界清楚,含有特征性的嗜酸性、粗的“绳状”胶原纤维。 |
ALT/WDLPS rarely involves the subcutaneous compartment but shows similar histologic appearances as atypical spindle cell lipomatous tumors, although a prominent spindle cell component is uncommon; IHC or fluorescence in situ hybridization (FISH) can be helpful to make this distinction. | ALT/WDLPS很少累及皮下,但其组织学表现与非典型梭形细胞脂肪瘤样肿瘤相似,尽管明显的梭形细胞成分并不常见;IHC或荧光原位杂交(FISH)有助于区分这两种疾病。 |
By IHC, the tumor cells in atypical spindle cell lipomatous tumors show variable expression of CD34, S100 protein, and desmin, whereas MDM2 and CDK4 are generally negative (rarely, weak or focal staining for the latter markers may be seen). | IHC显示,非典型梭形细胞脂肪瘤样肿瘤中的肿瘤细胞CD34、S100和desmin表达不定,而MDM2和CDK4通常为阴性(很少有弱染色或局灶染色)。 |
Spindle cell lipomas show diffuse CD34 expression, but by contrast are usually negative for desmin and S100 protein. | 梭形细胞脂肪瘤呈弥漫性CD34表达,但desmin和S100蛋白通常为阴性。 |
Notably, loss of nuclear RB1 expression is observed in 50% to 70% of atypical spindle cell lipomatous tumors, in keeping with chromosome 13q14 deletion, including the RB1 locus, which is also a consistent finding in spindle cell/pleomorphic lipomas. | 值得注意的是,在50%到70%的非典型梭形细胞脂肪瘤样肿瘤中观察到核RB1表达缺失,这与染色体13q14缺失一致,包括RB1基因位点,这也是梭形细胞/多形性脂肪瘤的一致发现。 |
MDM2 amplification is absent in atypical spindle cell lipomatous tumor, in distinction from ALT/WDLPS. | 与ALT/WDLPS不同,非典型梭形细胞脂肪瘤样肿瘤缺乏MDM2扩增。 |
Importantly, atypical spindle cell/pleomorphic lipomatous tumors pursue a benign clinical course with a low rate of nondestructive local recurrence (10% to 15%), lower than the risk for ALT/WDLPS; in contrast to ALT/WDLPS, atypical spindle cell lipomatous tumor has no potential to dedifferentiate. | 重要的是,非典型梭形细胞/多形性脂肪瘤样肿瘤呈一良性临床过程,其非破坏性局部复发率较低(10%-15%),低于ALT/WDLPS的风险;与ALT/WDLPS相比,非典型梭形细胞脂肪瘤样肿瘤没有去分化的潜力。 |
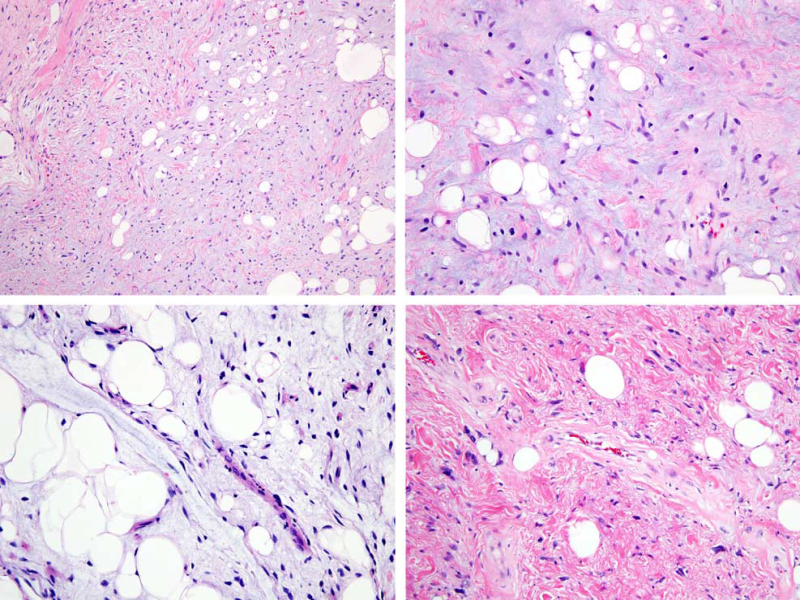
FIGURE 1. Atypical spindle cell lipomatous tumor. A, This tumor shows a variably collagenous to myxoid stroma and is dominated by spindle cells, in areas admixed with adipocytes. B, The tumor cells are relatively uniform, whereas the adipocytes show marked variation in size, including occasional lipoblasts. C, This tumor contains abundant myxoid stroma with prominent adipocytes and scattered spindle cells with hyperchromatic nuclei. D, This example shows dense collagenous stroma, notably pleomorphic and multinucleated cells, and occasional lipoblasts.
图1 非典型梭形细胞脂肪瘤样肿瘤:A、此肿瘤显示不同程度的胶原到黏液样间质,主要由梭形细胞组成,混合脂肪细胞。B、肿瘤细胞相对一致,而脂肪细胞大小明显不一,包括偶见的脂肪母细胞。C、此肿瘤含有丰富的黏液样间质,以脂肪细胞为主,可见散在的核深染梭形细胞。D、本例显示致密的胶原基质,显著的多形性和多核细胞,偶尔还有脂肪母细胞。
我要评论






共0条评论